Research
Background
Over the past two decades, technological advances have revealed that the three-dimensional organization of chromatin plays a crucial role in regulating gene expression.
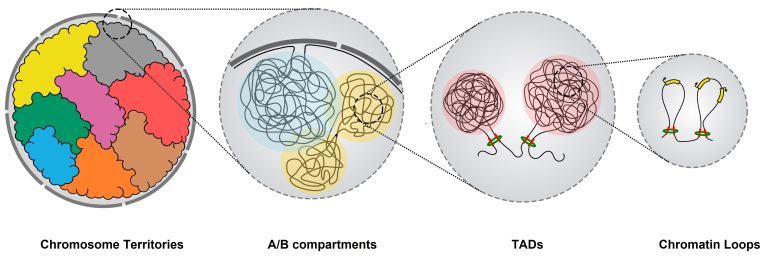
During interphase, the genome is arranged into a hierarchy of structural domains within the nucleus. At the largest scale, chromosomes occupy distinct regions known as chromosome territories, where gene activity is influenced by their proximity to nuclear structures associated with either gene repression or activation. At finer levels of organization, chromatin is divided into A and B compartments, representing transcriptionally active and inactive genomic environments. Within these compartments, the genome further organizes into topologically associating domains (TADs), regions that promote interactions between regulatory elements such as enhancers and promoters, enabling coordinated gene regulation. At the most local scale, chromatin loops create microenvironments that directly facilitate enhancer–promoter interactions and fine-tune gene expression.
Our research aims to understand how these structural and epigenetic features shape both normal and disease-associated transcriptional programs, particularly in the context of cardiac disease. To achieve this, we combine high-throughput genomic techniques (including Hi-C, ATAC-seq, ChIP-seq, RRBS, and RNA-seq) with advanced microscopy and animal models that replicate human heart disease.
Projects
Examination of the effect that cardiac stressors exert on chromatin topology. Heart failure is highly associated with comorbidities promoted by external factors like diet, smoking, or lack of exercise. In this project, we are studying how these stressors participate in the setting of cardiac pathology by reorganizing cardiac chromatin structure.
Development of novel approaches to treat cardiac disease through modulation of chromatin structure. Different epigenetic strategies have been used to treat cardiac disease. However, modification of genomic structure has never been tested. This work aims to develop novel approaches to treat and prevent cardiac pathology based on the direct 3D remodeling of the cardiac chromatin.
Integrative analysis of cardiac epigenetics in human heart failure. Cardiac disease is a costly condition affecting millions of people worldwide. This study uses human cardiac tissue to collectively analyze the influence of epigenetic factors (DNA methylation, histone marks, chromatin accessibility, and high-order chromatin structure) in triggering the onset of heart failure.